Used for total cyanide distillation, the
MICRO DIST system (Lachat
Instruments-Hach, Loveland, CO) was
granted National Primary Drinking
Water Regulations (NPDWR) approval
in 2003 and is expected to receive nationwide
Tier III approval for National Pollution
Discharge Elimination System
(NPDES) in October 2005.1 The
MICRO DIST (Figure 1) can be used
instead of the cumbersome, slow, and
error-prone large-scale glassware apparatus
described in U.S. EPA Method 335.22
and Standard Methods 4500-CN-C.3
This study evaluates the proposed distillation
for total cyanide in a variety of water
matrices with two flow injection analysis
systems as the determinative step.

Figure 1 - Samples and standards distilling simultaneously
in a MICRO DIST block.
As part of the study, interlaboratory and
intralaboratory tests were performed to
evaluate method performance. The
method detection limit, precision, and
recoveries of selected metal cyanide complexes
were evaluated. Samples containing
Prussian blue were distilled with the
MICRO DIST system and compared
with the U.S. EPA distillation to evaluate
the recovery of colloidal cyanides.
The sample pH, storage temperature,
holding time, and interference issues
were also investigated.
Experimental
Standards and environmental samples
were digested and distilled using the
MICRO DIST system and were evaluated
with a QuikChem automated ion
analyzer (Lachat Instruments-Hach) or
a flow injection analysis (FIA) instrument
equipped with gas diffusion separation
and amperometric detection. Samples
(6 mL each) were pipetted into a
sample tube and then acidified with 0.75
mL of magnesium chloride/sulfuric acid-releasing
solution. The samples were distilled
for 30 min at 120 °C in a special
heating block that fits the sample tube
closely, allowing the poorly conducting
polypropylene to be heated rapidly yet
locally. As the sample boiled the vapors
passed through a hydrophobic, porous
membrane. The vapors condensed above
the membrane to form a liquid pool over
the membrane that could not pass back
through into the sample.
A 1.0 M sodium hydroxide trapping solution
was added above the membrane to
convert hydrogen cyanide gas to the nonvolatile
cyanide anion. After the distillation,
the top of each tube was separated
from the bottom at the breakaway point
(Figure 2) and the distillate sample was
diluted to the original volume, equivalent
to 0.25 M NaOH, prior to analysis. The
cyanide in the distillates was analyzed with spectrophotometric or amperometric
detection systems. The two determinative
steps for the distillates are described below.
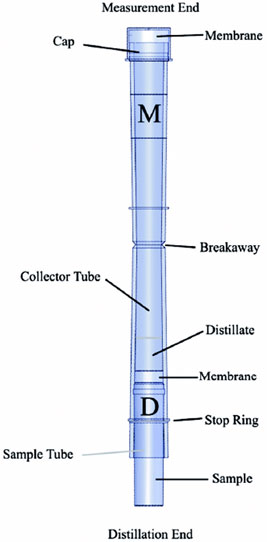
Figure 2 - MICRO DIST tube.
For spectrophotometric determinations,
the cyanide present in the 0.25 M NaOH
distillate was converted to cyanogen chloride
by reaction with chloramine-T, pyridine,
and barbituric acid to provide a red-colored
complex. The absorbance of this
complex was measured at 570 nm by
measuring the peak area resulting from
the sample. The peak area is proportional
to the concentration of
the cyanide in the sample.
For amperometric determinations,
the distilled samples were
analyzed as described in ASTM
Standard Test Method D 6888-
04.4 In this method, the distillates
are injected into an FIA system
and acidified on-line with
dilute sulfuric acid and bismuth
nitrate. The cyanide diffuses
through a gas diffusion membrane and is
captured into an alkaline acceptor stream.
The captured cyanide is sent to an amperometric
flow cell detector where the measured
anodic current is proportional to the
cyanide concentration.
Effect of method detection limit
The method detection limit (MDL) for
each determinative step was established
according to the U.S. EPA procedure.5
The MDL is defined by the U.S. EPA as
“the minimum concentration of a substance
that can be measured and reported
with 99% confidence that the analyte
concentration is greater than zero and is
determined from analysis of a sample in a
given matrix containing the analyte.”
A 5-μg/L CN– standard was distilled seven
times and analyzed by the spectrophotometric method shown in Figure 3. A 10-μg/L CN– standard was distilled seven
times and analyzed by the amperometric
method. The calculated MDL values were
<2 μg/L CN– for both determinative steps,
which is at least 10 times lower than the
detection limit published in U.S. EPA
Method 335.2.

Figure 3 - Method detection limit data for total cyanide with 5 μg/L CN–.
Recoveries of potassium ferricyanide [K3Fe(CN)6]
In the total cyanide method, many metal
complexes of cyanide are recovered. As a
recovery test, laboratory water samples
were fortified with K3Fe(CN)6 at 300 μg/L
CN– or 500 μg/L CN–. The metal cyanide
complex of [Fe(CN)6]3– was chosen
because of its relatively high stability constant
(43.6 log K at 25 °C) in order to
challenge the distillation.6 The mean
recovery was 305 μg/L CN– or 99.8%,
with an RSD of 1.5% (spectrophotometric
detection, n = 5) and a mean recovery
of 503 μg/L CN– or 101%, with an RSD
of 1.5% (amperometric detection, n = 4).
Three wastewater samples were fortified
with K3Fe(CN)6, distilled with the
MICRO DIST system, and then analyzed
with amperometric detection to
evaluate recovery. The samples were
believed to have a sulfide interference
with the U.S. EPA method; however,
the spike recoveries were found to be
acceptable with the evaluated procedure.
The data demonstrate that sulfide
can be mitigated with the bismuth
nitrate that is in the acidification solution
of ASTM D 6888-04 (see Table 1).
Table 1 - Fortified wastewater*

Quality control samples
A reference water sample from Environmental
Resource Associates (ERA,
Arvada, CO), lot no. 9959, was digested and distilled in duplicate. Its certified
concentration for total cyanide was 219
μg/L CN–. The observed concentration
with the spectrophotometric determination
was 195 μg/L CN–, which resulted in
an 88.9% recovery. Quality control data
are tabulated in Table 2 for samples that
were distilled with the MICRO DIST
system and then analyzed by amperometric
detection. All of the quality control
samples were within the acceptance
range for each QC sample.
Table 2 - Quality control summary for MICRO DIST followed by
amperometric detection

Tier III alternative test procedure (ATP) matrices and recovery results
In an effort to obtain nationwide approval
for drinking water and wastewater, a Tier
III ATP was conducted. This process
required that nine laboratories submit
recovery and performance data for nine
separate matrices. The recoveries for
matrix spikes and the relative percent difference
on their duplicates are shown in
Table 3. This study was conducted using
the MICRO DIST system followed by
spectrophotometric determinations.
Table 3 - U.S. EPA Tier III summary

Prussian blue recoveries
A Prussian blue recovery solution was prepared
by accurately weighing 11.0 mg of
the pigment, ferri-ferrocyanide (CAS
14038-43-8), into 1 L total volume
0.01 M sodium hydroxide, resulting in a
theoretical concentration of 6.00 mg/L as
CN–. When Prussian blue is subjected to
alkaline conditions of sample preservation,
it forms a brown-colored solution of
iron(III) hydroxide and ferrocyanide that
should return to blue upon acidification.
Tests were conducted to compare the
cyanide recoveries of the MICRO DIST
system with a traditional midi-distillation.
The stock solution of Prussian blue was
diluted to 300 μg/L as CN– in 0.01 M
sodium hydroxide (all of the Prussian blue
appeared to dissolve), and 10 replicates
were distilled with each method for total
cyanide. All distillates were analyzed with
ASTM D 6888-04 (gas diffusion separation
and amperometric detection). The
data are summarized in Table 4. In addition,
a manufacturing wastewater was fortified
with Prussian blue with a mean recovery of
82.2% using the MICRO DIST system.
Table 4 -Prussian blue recovery
study

During the study it was discovered that
temperature, pH, and storage time affected
the recovery of Prussian blue. Samples subjected
to longer storage time at pH 12
showed significantly improved recoveries.
If samples are suspected to contain Prussian
blue, it is imperative to preserve the samples
with sodium hydroxide to pH 12 upon
sample collection and allow the samples
sufficient time to reach room temperature
prior to analysis. For example, the recovery
of cyanide from Prussian blue in a sample
that was stored at 4 °C increased from 64%
to 84% by allowing the sample to warm to
room temperature prior to analysis.
Interference issues
Interference issues from sulfide (S2–),
nitrates/nitrites (NOx), and thiocyanate
(SCN–) could potentially cause false positive
or negative results if not mitigated.7
Sulfide is typically removed by adding lead
carbonate to the sample and/or to the
absorbing solution during the distillation;
however, the rapid loss of cyanide is possible
due to the formation of thiocyanate.8
The data from this study demonstrate that
sulfide can be removed by performing the
determinative step employing the sulfide
abatement procedure described in ASTM
D 6888-04 using bismuth nitrate in the
acidification reagent during flow injection
analysis followed by amperometric detection.
For spectrophotometric determinations,
lead carbonate can be added to the
absorbing solution after the distillation is
complete, followed by immediate filtration
to avoid the formation of thiocyanate.
Nitrate and nitrite in the presence of
thiocyanate or other compounds may
form HCN during the distillation. This
can be avoided by adding 0.25 mL 1 M
sulfamic acid solution to the sample during
the distillation step. However, up to
50 μg/L CN– positive interference was
observed in synthetic precious metals
mining wastewater samples with 15 mg/L
SCN– in the presence of 25 mg/L NH3 as
N and 25 mg/L NO3 as N. This interference
is likely due to the harsh conditions
of the distillation (high temperature and
acidic conditions) and also occurs with
U.S. EPA Method 335.2; therefore, it is
not recommended to use any distillation
method with this particular sample type.
Conclusion
The MICRO DIST system proved to be a
good alternative to conventional cyanide
distillations. The system uses 80 times less
sample, yet is capable of a detection limit that is 10 times lower than that of U.S.
EPA Method 335.2. The system takes
considerably less time to set up, and
throughput of samples is greatly increased
to approx. 21 distillations per hour. Since
the MICRO DIST tubes are single use,
there is essentially little or no cleanup.
Recoveries were adequate for all total
cyanide species evaluated during the study.
The method is applicable to a wide range
of sample matrices, including samples that
contain high levels of suspended solids or
colloidal cyanides. Interference issues can
be mitigated in most samples using the
techniques described in this article.
References
- Fed Reg Apr 6, 2004. Proposed rules, vol 69, no. 66.
- U.S. EPA Method 335.2. Cyanide Total, 1980.
- Method 4500-CN-C. Total Cyanide After Distillation. Standard Methods for the Examination of Water and Wastewater, 20 ed., APHA, 1998.
- ASTM D 6888-04. Standard Test Method for Available Cyanide With Ligand Displacement and Flow Injection Analysis Utilizing Gas Diffusion Separation and Amperometric Detection, 2004.
- Appendix B, 40 CFR Part 136. Definition and Procedure for the Determination of the Method Detection Limit, rev. 1.11, 1986.
- ASTM D 6696-01. Standard Guide for Understanding Cyanide Species, 2001.
- ASTM D 2036-98. Standard Test Method for Cyanides in Water, 1998.
- Wilmot JC, Solujic L, et al. Formation of thiocyanate during removal of sulfide as lead sulfide prior to cyanide determination. Analyst Jun 1996; 121:799–801.
Mr. Sebroski is Associate Scientist, Materials
Characterization, Bayer Material Science LLC,
100 Bayer Rd., Pittsburgh, PA 15205, U.S.A.;
tel.: 412-777-3420; fax: 412-777-7640, e-mail:
[email protected]. Ms.
Bogren is Program Manager, Lachat Instruments-Hach, 5600 Lindbergh Dr., Loveland,
CO 80538, U.S.A.; tel.: 970-663-1377; fax:
970-962-5610; e-mail: [email protected].